Research Topics
-
- Research
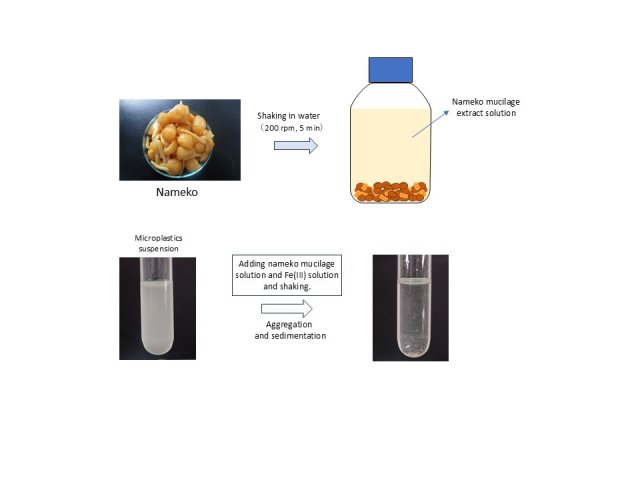
Nameko Mucilage: An Eco-Friendly Solution for Microplastic Pollution
-
- Research
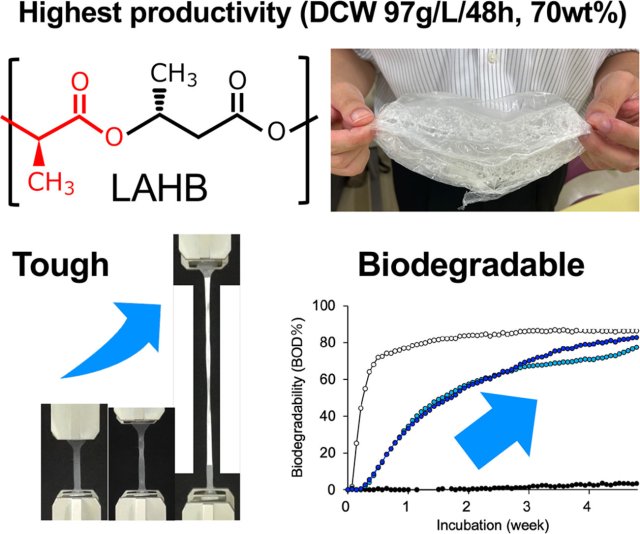
Reinforced Enzyme Expression Drives High Production of Durable Lactate-Based Polyester
-

- Global
- Research
Dr. Eugenio H. Otal wins a Poster Presentation prize at IAS International Conference on Energy Materials
-
- Research
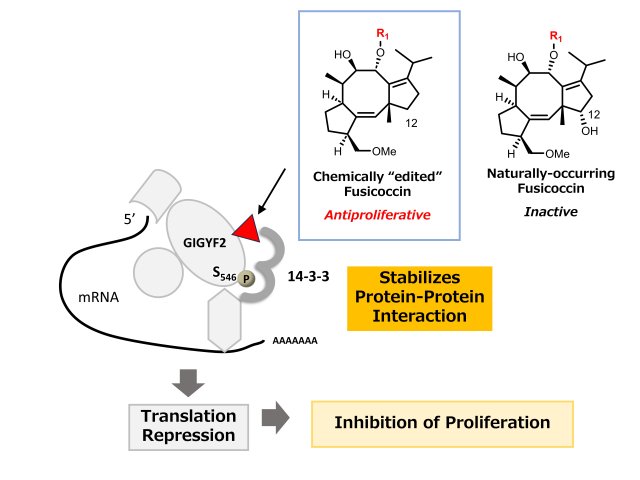
Chemically Edited Molecular Glue in Action: How 12-Deoxyfusicoccin Locks a Repressor
-
- Research
Efficacy of Immunosuppressive Regimens for Survival of Stem Cell-Derived Grafts
-

- Research
Ribosomal Engineering Creates "Super-Probiotic" Bacteria
-

- Collaborate
- Global
- Research
Institute for Aqua Regeneration and Faculty of Science and Technology, Thammasat University Sign Memorandum of Understanding
-

- Collaborate
- Global
- Research
Institute for Aqua Regeneration and Thai, VISTEC signed MoU
-

- Collaborate
- Global
- Research
Institute for Aqua Regeneration and Faculty of Engineering, KMUTT Sign Memorandum of Understanding
-
- Research
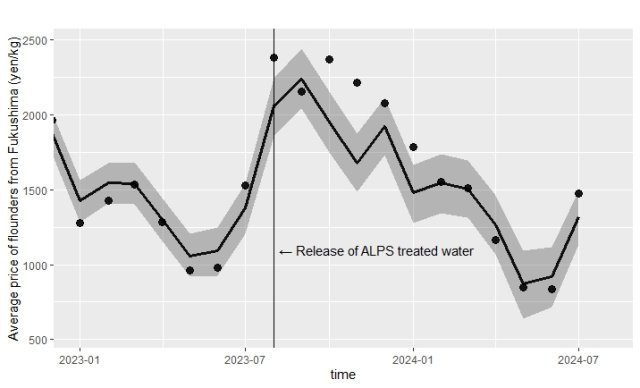
Shinshu University Finds No Price Drop in Fukushima Flounder After Treated-Water Discharge
-
- Research
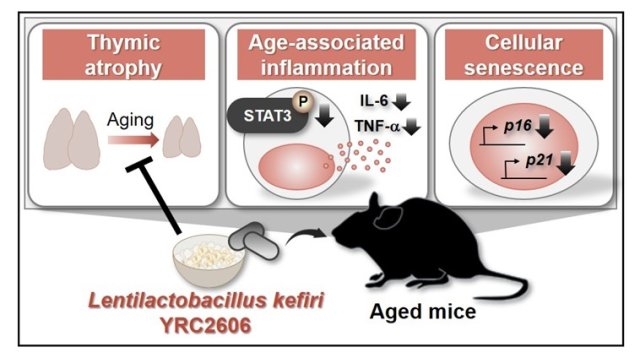
Can Certain Bacteria Regulate Aging of the Immune System and Its Related Alterations?
-

- Research
Probiotics and Prebiotics Offer Safer Alternatives to Antibiotics in Animal Agriculture
-

- Global
- Outreach
- Research
Global Spotlight on Shinshu University's Water Purifying Technology: "Shindai Crystal®" Featured in Japanese Government Video
-

- Research
Could Cardamom Seeds be a Potential Source of Antiviral Treatment?
-

- Outreach
- Research
Proposing the Future of Water and Energy at the Science Summit 2025 Held Alongside the UN General Assembly

